
Research in the lab focuses on both the causes and consequences of behavioral evolution. Using a colorful group of North American freshwater fish called darters, we ask the following questions:
Why does mate choice evolve over time? Why has male nuptial color pattern diversified in such spectacular ways in this lineage? How does the rate of behavioral divergence compare with the evolution of other reproductive barriers? Why are individuals attracted only to members of their own species?

(For that matter, ARE all individuals necessarily attracted only to members of their own species?) What is the relative contribution of male and female mate preferences to the maintenance of species boundaries?
Projects
Sex roles and the OSR
Cynthia F. O’Rourke, Ph.D. student
Background: “Sex roles” are defined as being either choosy or competitive, with females typically the choosy sex and males the competitive. Re-examination of these classic sex roles suggest that they may not be so rigid; rather, individuals may change their behavior in response to a changing environment.
Question: Is the degree to which individuals are choosy or competitive a plastic response to changes in the operational sex ratio (OSR)?

Phylogenetic analysis
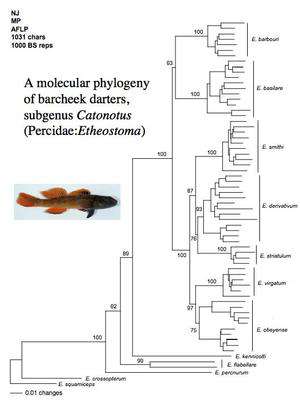
barcheek darters (subgenus Catonotus)
Reconstructing the evolutionary history of species is a fundamental component of research in my lab. First, I need a robust estimate of phylogeny to reveal phylogenetically (statistically) independent pairs of species on which to do analysis. Second, I use phylogeny estimates to reconstruct rates and geographical patterns of speciation. We use molecular markers to estimate phylogenies, including mitochondrial DNA sequences and amplified fragment length polymorphisms (AFLPs). AFLPs are particularly useful for resolving relationships among very closely related species. Future work will continue to use these molecular markers to estimate phylogenetic relationships within subgenera of darters.
Signal and sensory co-evolution
Jennifer M. Gumm, Postdoctoral associate
Background: One model of sexual selection posits that aspects of the environment can drive the evolution of male courtship signals indirectly, through their influence on female sensory systems.
Question: Do differences in male nuptial color correlate with differences in female visual sensitivity and the photic environment?
Methods: Male nuptial color is quantified for eighteen species of the subgenus Ulocentra (“snubnose” darters). Female visual sensitivity is determined using retinal microspectrophotometry, and differences in the photic (light) environment are quantified using spectral reflectance of background substrate at spawning sites in the stream.
Collecting
It’s a tough job, but someone’s gotta hang out in Appalachian streams on cool sunny spring days and catch fish.


Mate choice and nuptial color

Tory H. Williams, Ph.D. student
Background: The elaborate breeding colors displayed by males in many animals are thought to pose a significant barrier to interbreeding among species, with females responding preferentially to the colors of conspecific males. This assumption is rarely tested explicitly.
Question: Are species boundaries maintained by female preference for conspecific male nuptial color?
Methods: 1). Females are offered a choice between painted models designed to imitate conspecific and heterospecific color. 2). Premating and postmating reproductive barriers are quantified to determine whether behavioral isolation forms the strongest barrier to interbreeding.
The accumulation of reproductive isolation
Michael D. Martin, Ph.D. student

Background: Behavioral isolation is the reduction in interbreeding between species due to differences in mating signals and preferences. Behavioral isolation is thought to evolve rapidly among species with strong sexual selection. Male darters appear to experience strong sexual selection for nuptial coloration.
Question: Does behavioral isolation evolve faster than other reproductive barriers to gene flow? Is behavioral isolation best explained by differences in male nuptial color and/or the environment?
Methods: Multiple pairs of species are examined for multiple reproductive barriers to determine whether behavioral isolation evolves first. Indices developed to quantify environmental and male color differences between species are compared to the strength of behavioral isolation.
Current Members
 Cynthia O’Rourke
Cynthia O’Rourke
Graduate Student: PhD Biological Sciences
 Tory Williams
Tory Williams
Graduate Student: PhD Biological Sciences
 Michael Martin
Michael Martin
Graduate Student: PhD Biological Sciences
 Tracy Smith
Tracy Smith
Graduate Student: PhD Biological Sciences
 Patrick Ciccotto
Patrick Ciccotto
Graduate Student: PhD Biological Sciences
 Matt Wong
Matt Wong
Undergraduate Researcher
Former Members
|
Rebecca Lampe
Laboratory technician
Rebecca ran the fish lab and is now pursuing her Master’s degree at University of California-Santa Barbara.
|
 |
|
Sarah Ogden
Research technician
|
 |
|
Alexandria Scott
Undergraduate Researcher
|
 |